UCAN CURE – Precision Decisions for Childhood Arthritis
Project Overview – Integrated Thematic Activities
The Health Challenge
Childhood arthritis is devastating and one of the most common chronic diseases affecting Canadian children. Children suffer from daily pain and stiffness, severely impacting their quality of life and participation. Uncontrolled joint inflammation results in irreversible damage with growth abnormalities and deformities requiring early joint replacement.
Although expensive ($15,000–$416,000/year per child), powerful new biologic therapies targeting inflammatory mediators dramatically improve outcomes. In Canada, biologics can only be accessed after failing traditional therapies, resulting in critical delays in accessing effective drugs, exposure to unnecessary risks, and millions wasted in ineffective treatments. Recent data show that early short-term biologic therapy, in children with the most severe subtype of arthritis, results in rapid and long-lasting disease control after only three months of treatment, preventing destructive arthritis, altering biology and possibly “curing” arthritis.
Our project will identify the children who will benefit from this paradigm-changing approach.
The UCAN Research Proposal
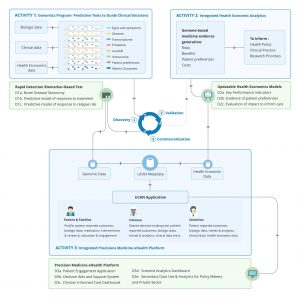
UCAN CURE will deliver a genomics-based predictive tool to determine when and how specific biologic agents should be safely and effectively used and when they can be discontinued, thus improving outcomes, limiting risk, and reducing socioeconomic burden. Work by our team has already established that patients and treatment response can be meaningfully classified using approaches that integrate comprehensive genomic and clinical data in a predictive tool to guide therapy.
Deliverables
| Current Challenges in Childhood Arthritis | UCAN CURE Deliverable |
|---|---|
| One in three children with arthritis need biologics, selected in a trial-and-error-fashion, prescribed without end-dates. | Deliverable 1: Rapid detection biomarker-based test for clinicians to predict risk for long-term disability, enable rapid selection of targeted biologic therapies and safe discontinuation of drugs |
| Lack of data to inform health policy decisionmakers on cost effectiveness, patient preferences, and affordability. | Deliverable 2: Updatable model of the risks, benefits and costs associated with genomics-based medicine to inform health-policy decision makers about cost effectiveness, patient preferences and affordability. |
| Inadequate collaboration among consumers, health-care providers, researchers, policymakers and other end-users leading to suboptimal clinical care and inappropriate policies. | Deliverable 3: A precision medicine eHealth platform to enable inclusive collaboration between consumers, health-care providers and researchers to transform clinical care and discovery. |
UCAN CURE will build on our previous work and bring genomic science and economic modelling (integrated GE3LS component) to the next level by combining individualized treatment pathways with models for cost-effectiveness, affordability and patient preferences – delivering updatable models of risks, benefits and costs associated with genomics-based care for childhood arthritis. Our state-of-the-art eHealth platform will move patient engagement and information sharing into the 21st century, supporting clinical management and encompassing the entire patient experience from eConsent to multi-directional information sharing between researchers, clinicians, policy-makers, patients and families to the gamification of the research pipeline and early education of the future generation of genomic researchers. Our group is at the forefront of data collection and international collaboration, leading national and international translational research networks. As all Canadian paediatric rheumatologists are collaborators, there is an explicit commitment, together with our international partners, to the UCAN CURE mission of real-life integration of innovative precision medicine strategies into care provision, forever transforming the care of children with arthritis in Canada and beyond.
